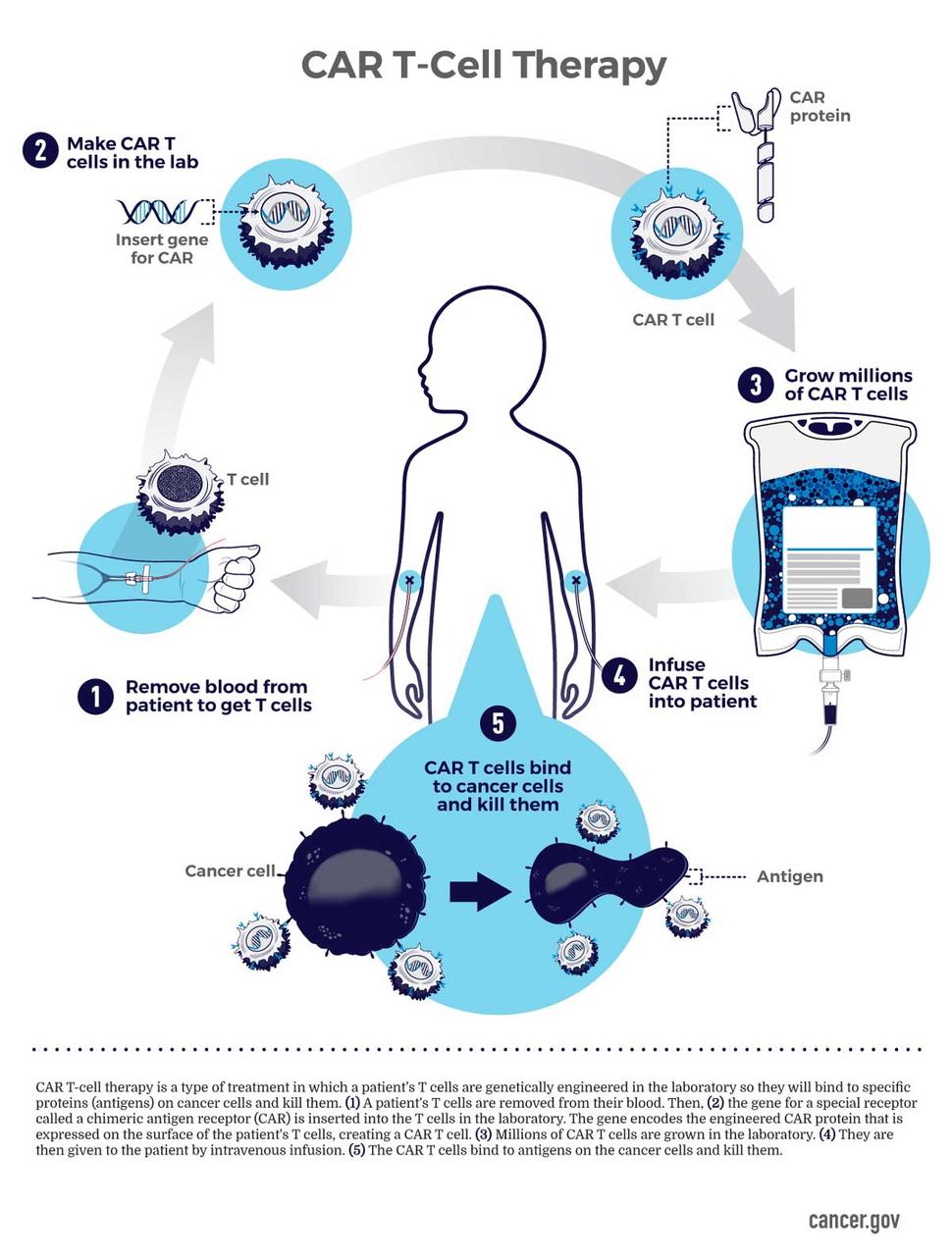
Car T Cell Therapy Infographic Nci Car t cell–mediated responses are further enhanced by adding intracellular costimulatory domains (e.g., cd28, 4 1bb), which cause significant car t cell expansion and may increase the lifespan of these cells in the recipient. car t cell therapy indications for pediatric cancer. Car t for aml is still in the early stages, but studies are ongoing. researchers are also looking into the potential use of car t cell therapy to treat solid tumors, which have been more challenging to treat with immunotherapy because the tumors find ways of excluding t cells and preventing them from entering and killing the tumor. dr.
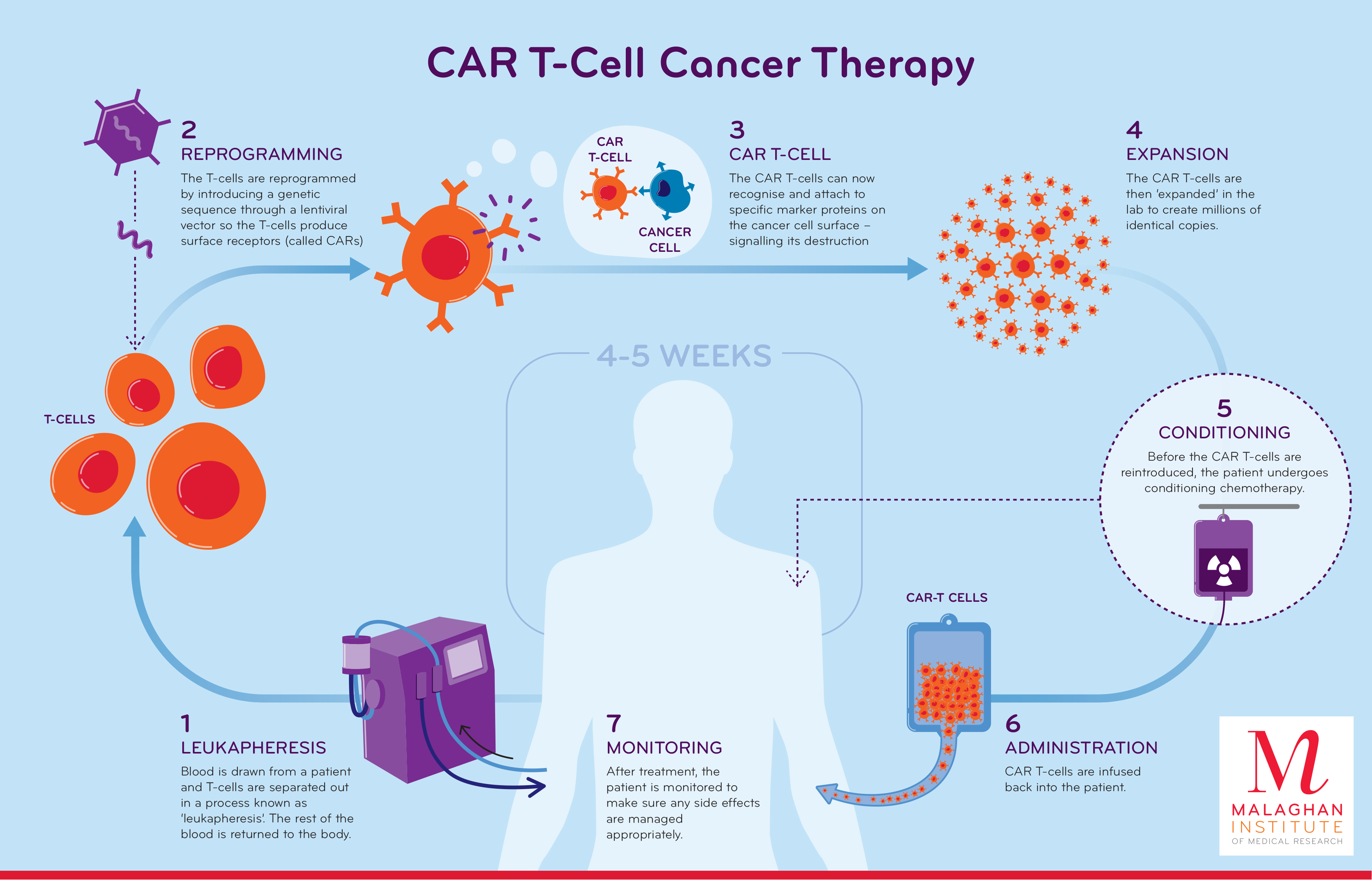
Car T Cell Therapy Malaghan After 5 p.m., call 212 639 2000. ask for the doctor covering bone marrow transplant. if you’re having an outpatient car t cell infusion, between 9 a.m. and 7 p.m., call the cellular immunotherapy unit at 646 608 3150. after 7 p.m., call 212 639 2000. ask for the doctor covering bone marrow transplant. A multidisciplinary team's recommendations help in the recognition, workup, evaluation, and management of the most common chimeric antigen receptor (car) t cell–related toxicities. in november 2021, the american society of clinical oncology (asco) released a new guideline entitled “management of immune related adverse events in patients. Make a referral. mayo clinic's car t cell therapy program offers a new cancer immunotherapy that involves genetically modifying t cells to activate the immune system to recognize and destroy certain cancers. chimeric antigen receptor (car) t cell therapy, also known as car t cell therapy, was approved by the food and drug administration in. Our appointment staff will work to find the specialist who can best address your questions and needs. be sure to mention that you are interested in car t cell therapy to ensure your request is routed correctly. minnesota: 507 284 5096. arizona: 480 342 4800. florida: 904 956 3309.
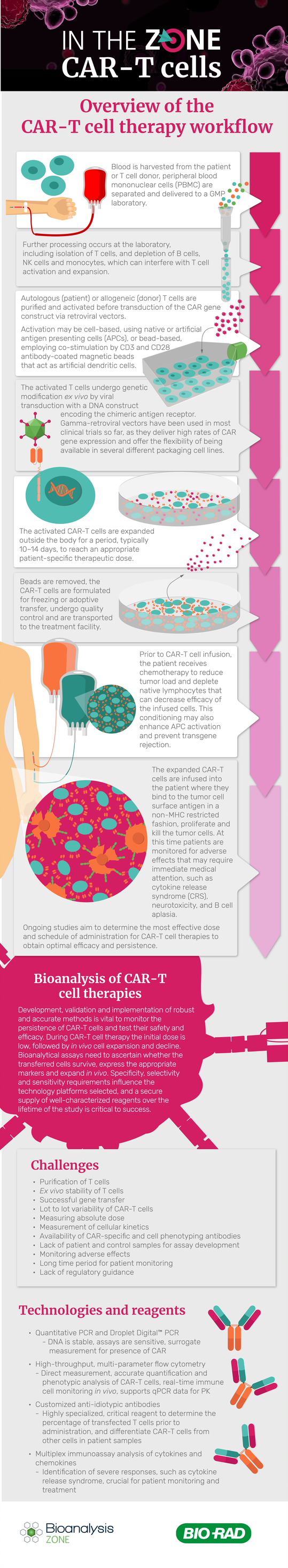
The Car T Cell Therapy Workflow Bioanalysis Zone Make a referral. mayo clinic's car t cell therapy program offers a new cancer immunotherapy that involves genetically modifying t cells to activate the immune system to recognize and destroy certain cancers. chimeric antigen receptor (car) t cell therapy, also known as car t cell therapy, was approved by the food and drug administration in. Our appointment staff will work to find the specialist who can best address your questions and needs. be sure to mention that you are interested in car t cell therapy to ensure your request is routed correctly. minnesota: 507 284 5096. arizona: 480 342 4800. florida: 904 956 3309. St. jude children's research hospital: “collecting cells for car t cell therapy.” memorial sloan kettering cancer center: “car t cell therapy: a guide for adult patients & caregivers. Car (chimeric antigen receptor) t cell therapy is a new form of immunotherapy. it uses altered t cells to target cancer cells. the immune system consists of many cells and organs. they almost always protect the body from infection and cancer. an important part of the immune system is t cells. t cells can hunt down and destroy abnormal cells.