A Xps Of The Cu 2p Spectrum And B The Peak Fitted Cu Lmm Auger Figure 7 demonstrates the oxidation states of cu in the aggregates by x ray photoelectron spectroscopy (xps). two binding energy peaks at 932.4 and 952.1 ev were observed, which were attributed to. The spectrum corresponding to 1 minute shows characteristic features associated with a cu 1 ; these are a fine main photoemission peak centered at 932.63 ev, the same binding energy for metallic copper, and a weak satellite peak located at 946.54 ev, the previously described m3 peak.

Copper Harwellxps Guru X ray induced structural evolution on the surface of na glass. x ray irradiation induced remarkable spectral changes in cu 2p and cu lmm spectra of na glass. in cu 2p spectra, a new peak (cu x band) appeared at 931.5 ev accompanied with the decrease of cu 2 and cu peaks at 935.4 and 932.5 ev, respectively. In the peak fit of cu lmm, shown in figure 2 a, there are two peaks at 568.3 ev (cu metal; peak 0) and 570.0 ev (cu 2 o; peak 1) and three other peaks at 573.1, 567.1, and 565.2 ev that represent. Download scientific diagram | a) cu 2p xps, b) cu lmm auger, and c) i 3d xps spectra of cu, cui, and cu cui electrodes after co 2 rr. from publication: high‐rate co2 electroreduction to c2. Auger parameter. it is defined as the difference in be between 2 chemical states. this depends on the change in core electron level energy and the change in intra and extra atomic relaxation energies. for a 1s (k) electron. Δe b(k) = Δε(k) Δr(k ) {Δ in relaxation e for singly ionized state} for kll auger process, the Δ ke between.

Quasi In Situ Cu Auger Lmm Xps Spectra Of Cu Co C U Br And Cu Download scientific diagram | a) cu 2p xps, b) cu lmm auger, and c) i 3d xps spectra of cu, cui, and cu cui electrodes after co 2 rr. from publication: high‐rate co2 electroreduction to c2. Auger parameter. it is defined as the difference in be between 2 chemical states. this depends on the change in core electron level energy and the change in intra and extra atomic relaxation energies. for a 1s (k) electron. Δe b(k) = Δε(k) Δr(k ) {Δ in relaxation e for singly ionized state} for kll auger process, the Δ ke between. Goh et al.[2] have shown (in their figure 8) the distinctly different peak shapes of the x ray generated auger lmm spectra for copper as the metal, cu 2 s and cus. they also note the distinctive cu l 3 m 4,5 m 4,5 peak at 916.5 ev for cu 2 o. poulston et al.[3], in their study of surface oxidation and reduction of cu 2 o and cuo, have used both. They also note the distinctive cu l 3 m 4,5 m 4,5 peak at 916.5 ev for cu 2 o. poulston et al. [64], in their study of surface oxidation and reduction of cu 2 o and cuo, have used both the cu lmm and the auger parameter to distinguish cu(0), cu(i) and cu(ii). these parameters are very useful for the identification of the different states.
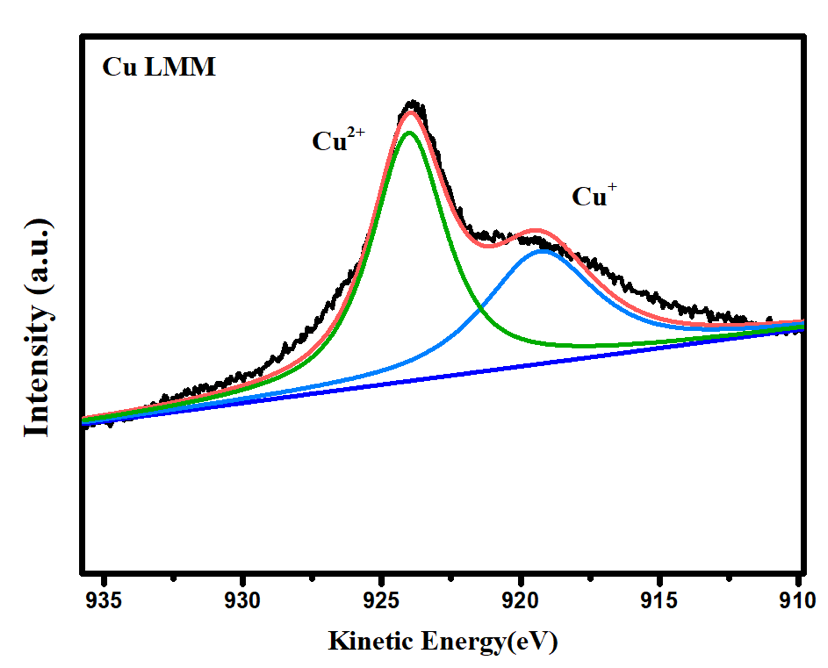
е ідєћcuзљ е ѓзґ д жђѓxpsгђѓlmmпјњlvvпјџ з еѕ ењ е ґй з Goh et al.[2] have shown (in their figure 8) the distinctly different peak shapes of the x ray generated auger lmm spectra for copper as the metal, cu 2 s and cus. they also note the distinctive cu l 3 m 4,5 m 4,5 peak at 916.5 ev for cu 2 o. poulston et al.[3], in their study of surface oxidation and reduction of cu 2 o and cuo, have used both. They also note the distinctive cu l 3 m 4,5 m 4,5 peak at 916.5 ev for cu 2 o. poulston et al. [64], in their study of surface oxidation and reduction of cu 2 o and cuo, have used both the cu lmm and the auger parameter to distinguish cu(0), cu(i) and cu(ii). these parameters are very useful for the identification of the different states.
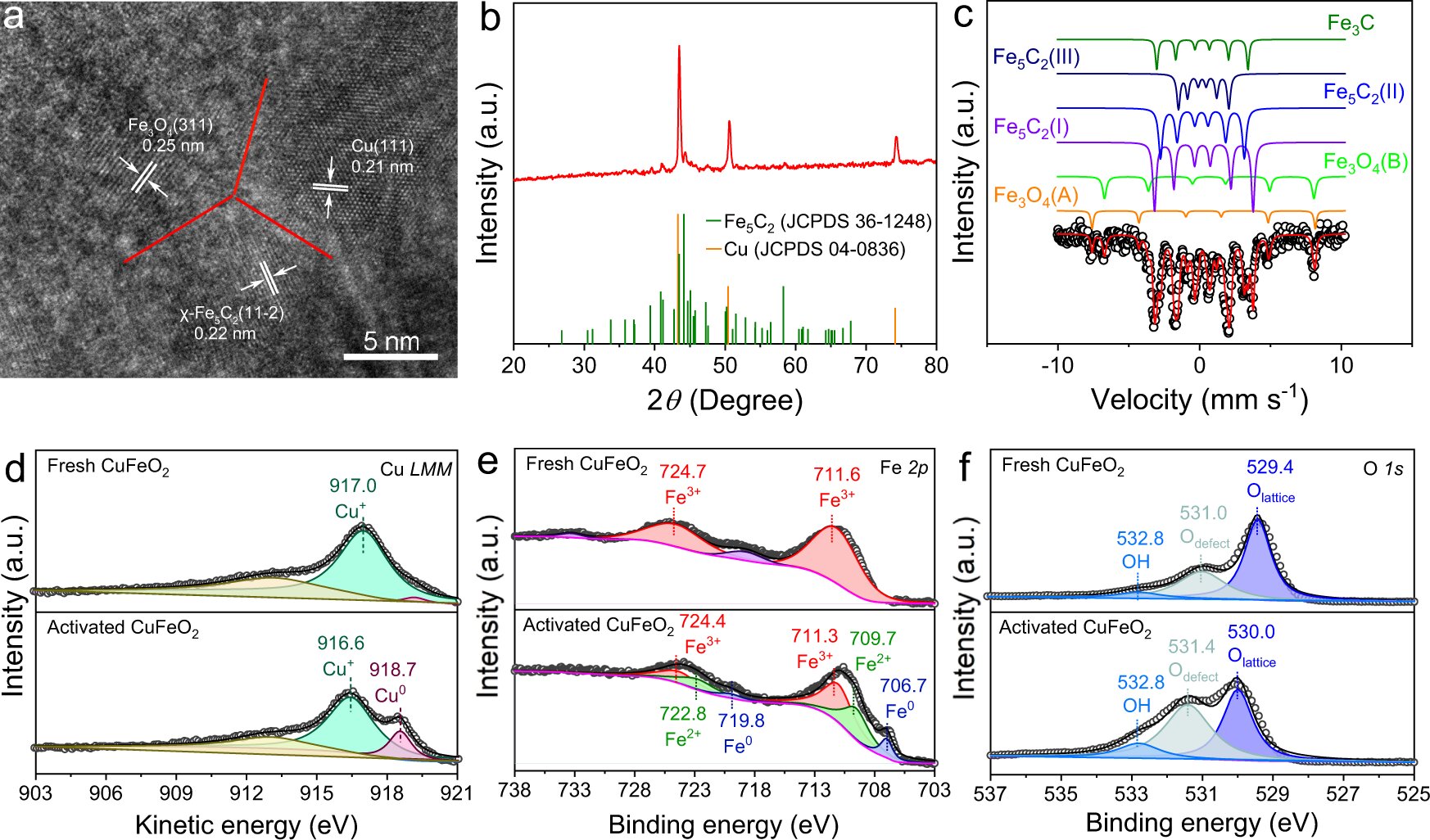
Cu Fe Catalyst Facilitates Ambient Pressure Conversion Of Co2 Into Long