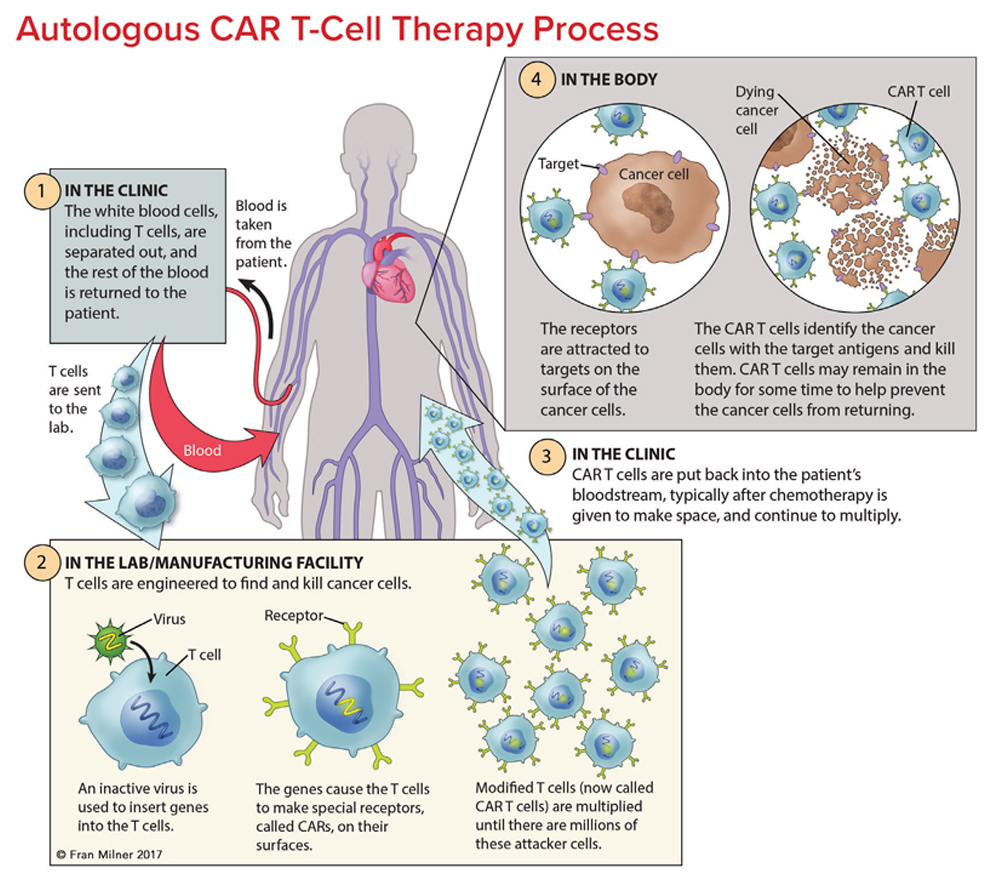
Chimeric Antigen Receptor Car T Cell Therapy 2022 Chimeric antigen receptor (car) t cell therapy represents a major advancement in personalized cancer treatment. in this strategy, a patient's own t cells are genetically engineered to express a synthetic receptor that binds a tumor antigen. car t cells are then expanded for clinical use and infused back into the patient's body to attack and. Ellebrecht, c. t. et al. reengineering chimeric antigen receptor t cells for targeted therapy of autoimmune disease. science 353 , 179–184 (2016). article cas pubmed pubmed central google scholar.

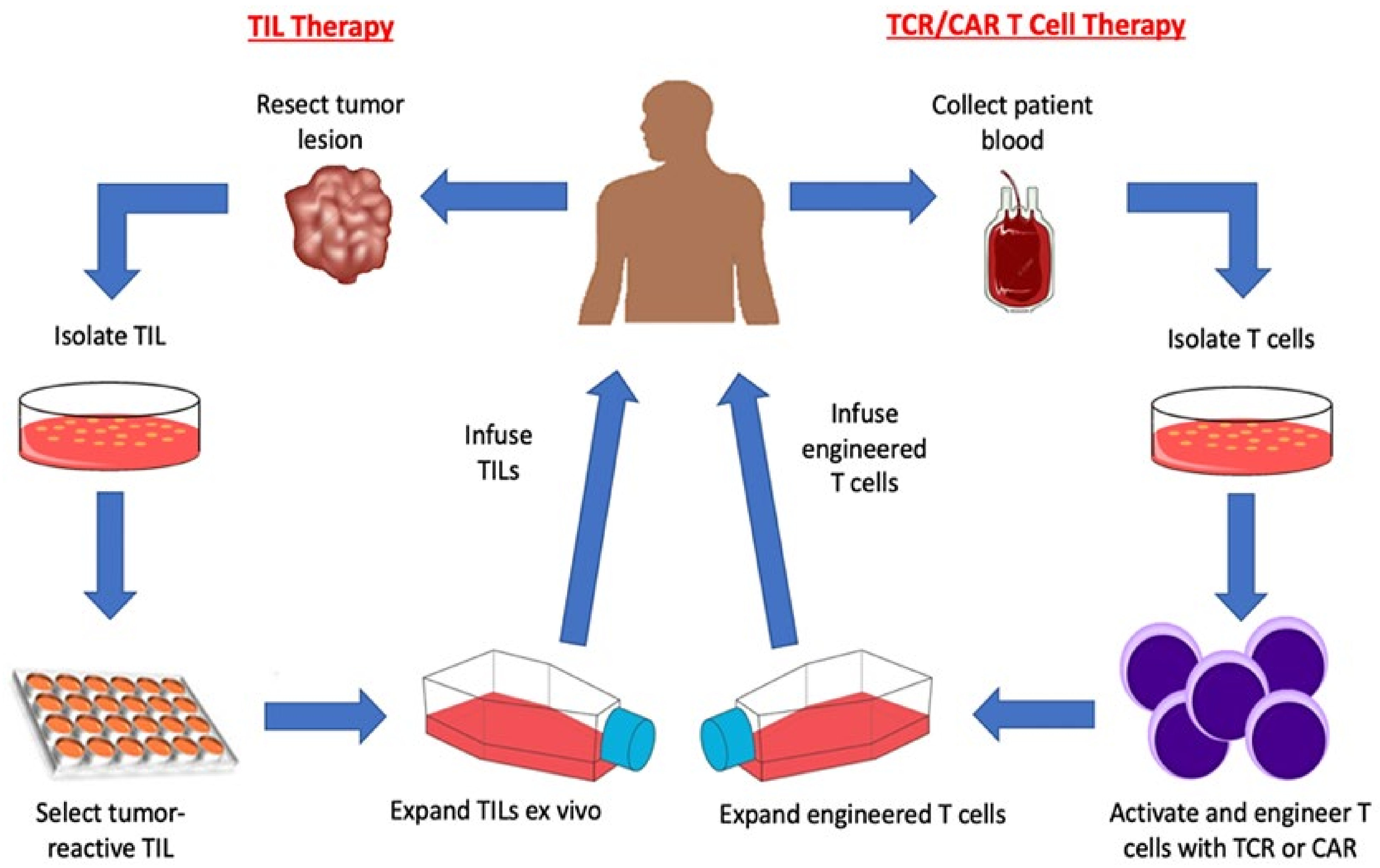
Adoptive T Cell Therapy And Chimeric Antigen Receptor Car Design Adoptive t cell therapy using chimeric antigen receptor (car) t cells is an alternative strategy for cancer therapy with high efficacy in hematological malignancies. Chimeric antigen receptor (car) t cells are highly effective at targeting and eliminating cells of the b cell lineage. car t cell therapy has become a standard of care treatment for patients with. Fig. 1. schematic overview of the processes for adoptive cell therapy (act) of tumor infiltrating lymphocytes (til), act with t cell receptor (tcr) gene therapy and act with chimeric antigen receptor (car) modified t cells. in act with til, tumor resident t cells are isolated and expanded ex vivo after surgical resection of the tumor. Introduction. chimeric antigen receptor (car) t cell therapy is a new class of adoptive cellular immunotherapy for cancer treatment involving ex vivo genetic manipulation of t cells, using either lentiviral or retroviral vectors or nonviral gene transfer systems to express engineered cars specific for particular tumor targets. 1,2 these reprogrammed car t cells are then infused into the.
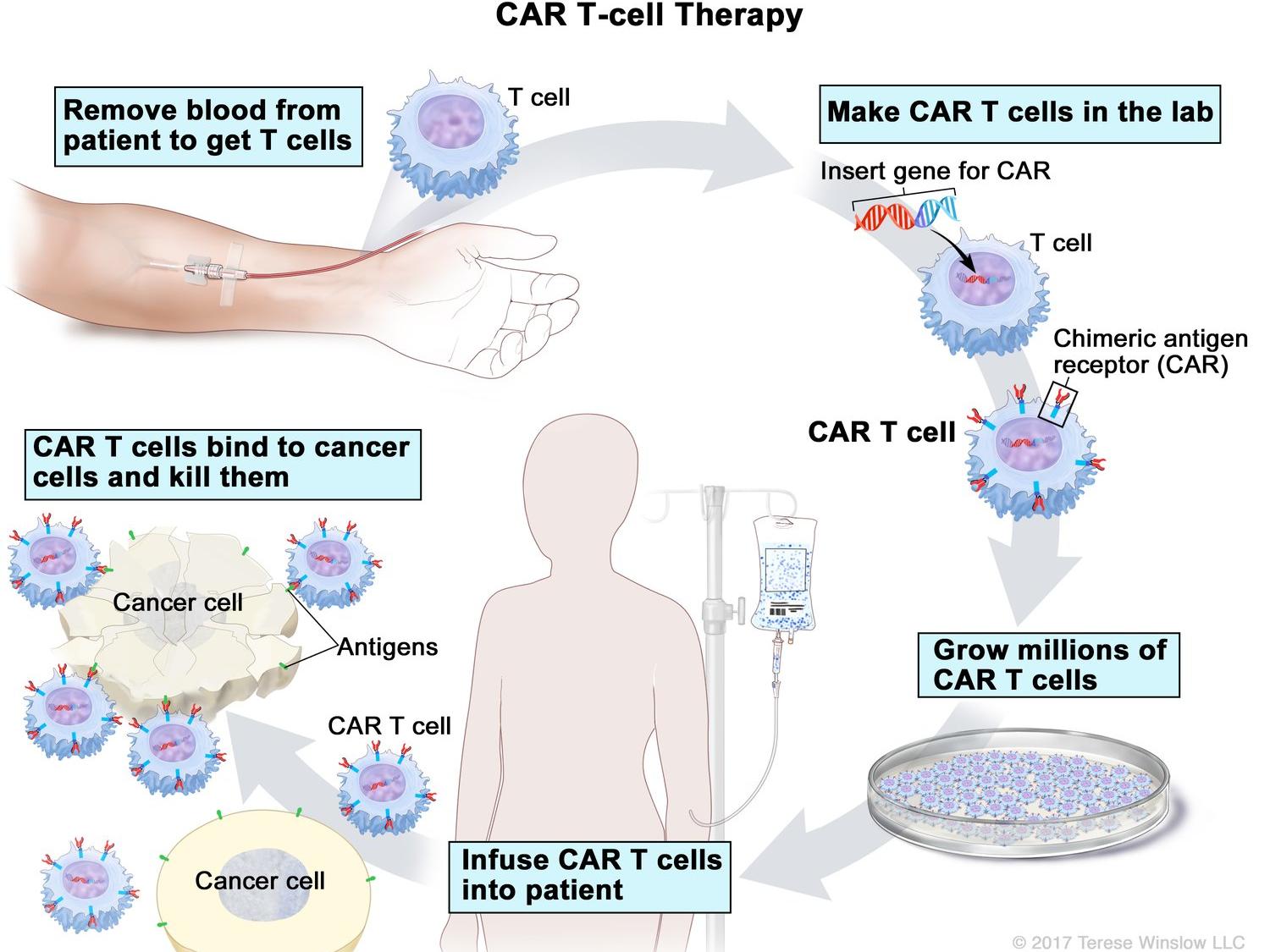
T Cell Transfer Therapy Immunotherapy Nci Fig. 1. schematic overview of the processes for adoptive cell therapy (act) of tumor infiltrating lymphocytes (til), act with t cell receptor (tcr) gene therapy and act with chimeric antigen receptor (car) modified t cells. in act with til, tumor resident t cells are isolated and expanded ex vivo after surgical resection of the tumor. Introduction. chimeric antigen receptor (car) t cell therapy is a new class of adoptive cellular immunotherapy for cancer treatment involving ex vivo genetic manipulation of t cells, using either lentiviral or retroviral vectors or nonviral gene transfer systems to express engineered cars specific for particular tumor targets. 1,2 these reprogrammed car t cells are then infused into the. Nevertheless, after years of painstaking research, car t cell therapies have entered the mainstream of cancer treatment, said steven rosenberg, m.d., ph.d., chief of the surgery branch in nci's center for cancer research (ccr), an immunotherapy and car t cell therapy pioneer. " [car t cells] are now widely available in the united states and. Chimeric antigen receptor t cell (car t) therapy is a form of immunotherapy that has enabled patients with previously incurable hematological cancers to go into remission. currently marketed car t therapies include yescarta, tecartus, kymriah, breyanzi, carvykti, and abecma. indications are acute lymphoblastic leukemia, large b cell lymphoma.
Vaccines Free Full Text Genetic Modification Of T Cells For The Nevertheless, after years of painstaking research, car t cell therapies have entered the mainstream of cancer treatment, said steven rosenberg, m.d., ph.d., chief of the surgery branch in nci's center for cancer research (ccr), an immunotherapy and car t cell therapy pioneer. " [car t cells] are now widely available in the united states and. Chimeric antigen receptor t cell (car t) therapy is a form of immunotherapy that has enabled patients with previously incurable hematological cancers to go into remission. currently marketed car t therapies include yescarta, tecartus, kymriah, breyanzi, carvykti, and abecma. indications are acute lymphoblastic leukemia, large b cell lymphoma.