
Chimeric Antigen Receptor Car T Cell Therapy Oncology Jama In the last decade, anti cd19 car t cell therapy has led to a treatment paradigm shift for b cell non hodgkin lymphomas, first with the approval for relapsed refractory (r r) large b cell lymphomas and subsequently for r r mantle cell and follicular lymphoma. many efforts are continuously being made to extend the therapeutic setting in the lymphoma field. several reports are supporting the. Simple summary. glioblastoma (gbm) is a highly aggressive brain tumor with low survival rates and limited treatment options. this review explores the potential of chimeric antigen receptor (car) t cell immunotherapy as a promising approach for treating gbm. while car t cell therapy has shown success in blood malignancies, it faces challenges.
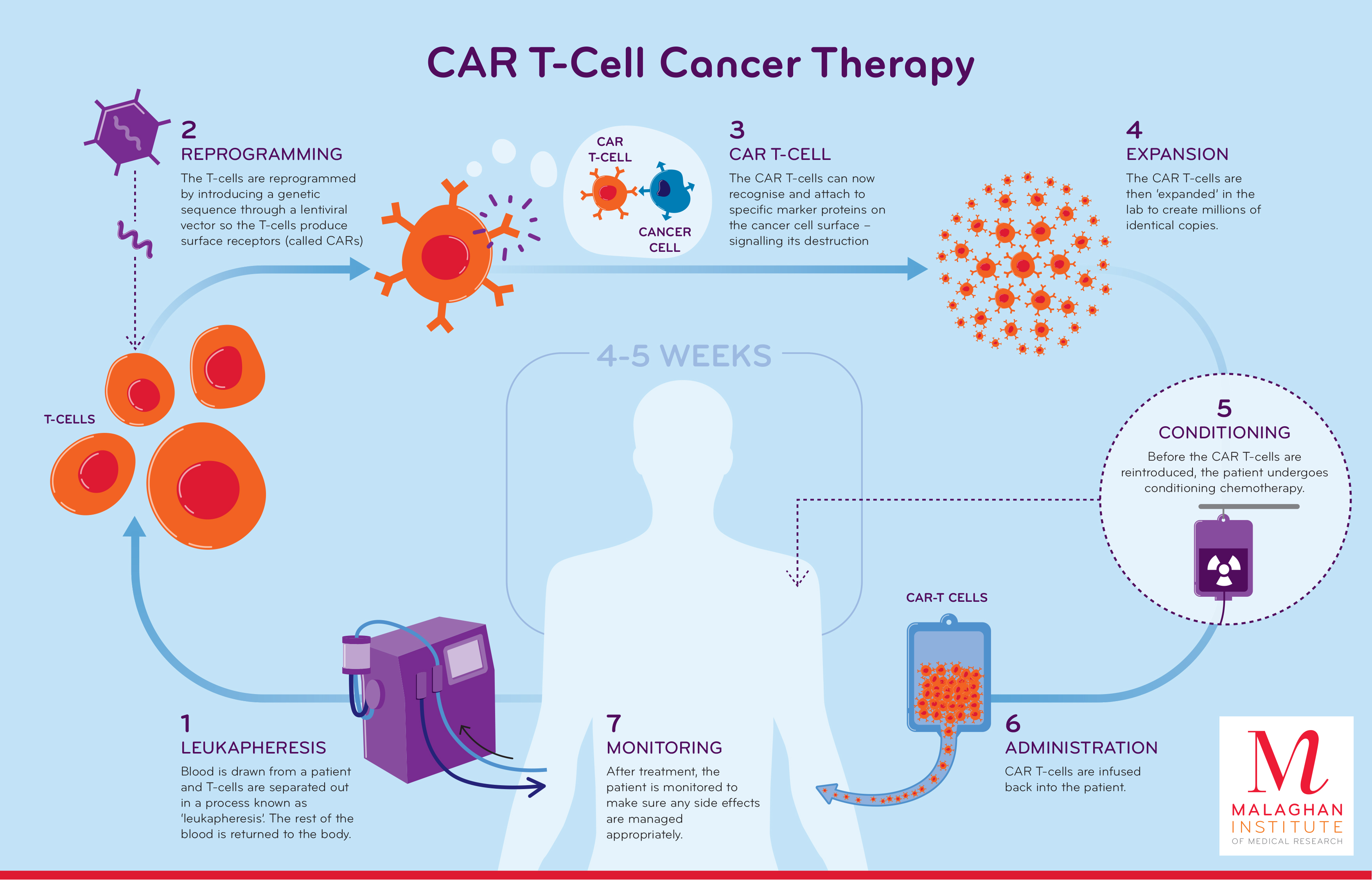
Car T Cell Therapy Chimeric antigen receptors (car) t cell therapy represents the most important innovation in onco hematology in recent years. the progress achieved in the management of complications and the latest generations of car t cells have made it possible to anticipate in second line the indication of this type of treatment in large b cell lymphoma. while some types of b cell lymphomas and b cell acute. Chimeric antigen receptor (car) t cell therapy utilizes engineered t cells to target specific tumor antigens, demonstrating high efficacy and gaining fda approval in some hematological malignancies. beyond oncology, it has recently also shown promise in treating autoimmune diseases. despite its successes, this therapy faces challenges with. Chimeric antigen receptor t cells have revolutionized the treatment of hematological malignancies during the past five years, boasting impressive response rates and durable remissions for patients who previously had no viable options. in this review, we provide a brief historical overview of their development. we focus on the practical aspects of a patient’s journey through this treatment. Editorial. chimeric antigen receptor t cell (car t) therapy is a form of immunotherapy that has enabled patients with previously incurable hematological cancers to go into remission. currently marketed car t therapies include yescarta, tecartus, kymriah, breyanzi, carvykti, and abecma. indications are acute lymphoblastic leukemia, large b cell.
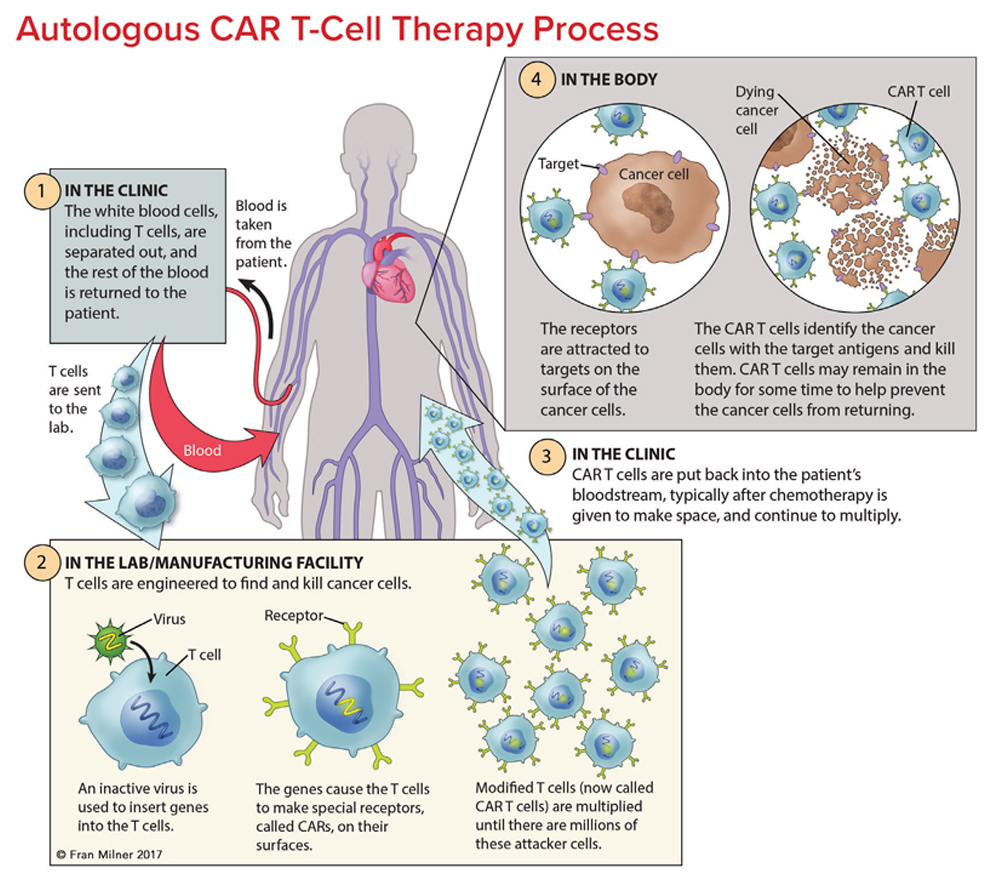
Chimeric Antigen Receptor Car T Cell Therapy 2022 Chimeric antigen receptor t cells have revolutionized the treatment of hematological malignancies during the past five years, boasting impressive response rates and durable remissions for patients who previously had no viable options. in this review, we provide a brief historical overview of their development. we focus on the practical aspects of a patient’s journey through this treatment. Editorial. chimeric antigen receptor t cell (car t) therapy is a form of immunotherapy that has enabled patients with previously incurable hematological cancers to go into remission. currently marketed car t therapies include yescarta, tecartus, kymriah, breyanzi, carvykti, and abecma. indications are acute lymphoblastic leukemia, large b cell. 22. martin t, usmani sz, berdeja jg, agha m, cohen ad, hari p, et al. ciltacabtagene autoleucel, an anti b cell maturation antigen chimeric antigen receptor t cell therapy, for relapsed refractory multiple myeloma: cartitude 1 2 year follow up. j clin oncol. (2023) 41:1265–74. doi: 10.1200 jco.22.00842. Although 9 of 10 patients were successfully treated, t cell leukemia developed in 4 of the 9 several years after gene therapy. 70 in more than 1000 patients infused with t cell receptors or car.
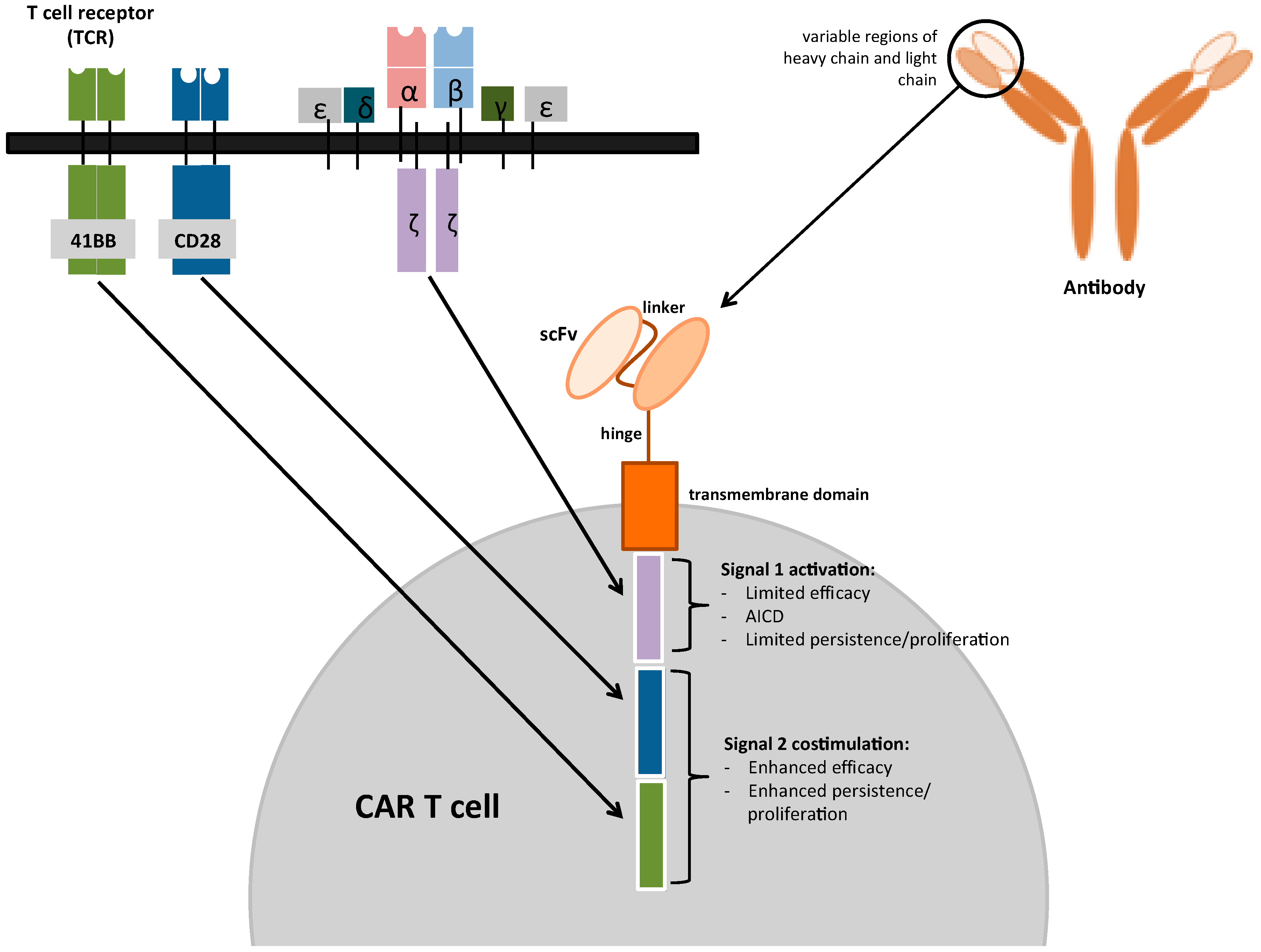
Mesothelioma Is It Rare The Panorama Of Different Faces Of Mesothelial 22. martin t, usmani sz, berdeja jg, agha m, cohen ad, hari p, et al. ciltacabtagene autoleucel, an anti b cell maturation antigen chimeric antigen receptor t cell therapy, for relapsed refractory multiple myeloma: cartitude 1 2 year follow up. j clin oncol. (2023) 41:1265–74. doi: 10.1200 jco.22.00842. Although 9 of 10 patients were successfully treated, t cell leukemia developed in 4 of the 9 several years after gene therapy. 70 in more than 1000 patients infused with t cell receptors or car.