
Is Bio Distribution Study Necessary For Car T Therapy вђ Creative Chimeric antigen receptors (cars) are recombinant receptors for antigens which redirect the specificity and function of t lymphocytes and or other immune cells in a single molecule. the concept of using cars in cancer immunotherapy is that cars, which are programmed targeting tumor associated antigens, can be replicated rapidly and homogeneously. direct infusion of these armed tumor targeting. In november 2021, the american society of clinical oncology (asco) released a new guideline entitled “management of immune related adverse events in patients treated with chimeric antigen receptor t cell therapy.” 1 the guideline, created by a multidisciplinary panel of medical oncology, neurology, hematology, emergency medicine, nursing, trialist, and advocacy experts, was developed.

Chimeric Antigen Receptor T Cell Therapy For Cancer Clinical Car t cells: engineering patients’ immune cells to treat their cancers. enlarge. co stimulatory signaling domains have been added to newer generations of car t cells to improve their ability to produce more t cells after infusion and survive longer in the circulation. credit: brentjens r, et al. “driving car t cells forward.”. Car t cell therapy is an individualized cell based technique that involves removing some of your own white blood cells, including t cells. to make car t cells, the collected t cells are genetically treated in the lab to produce special receptors called chimeric antigen receptors, or cars. these cars allow the t cells to recognize an antigen (or. Chimeric antigen receptor (car) t cells are highly effective at targeting and eliminating cells of the b cell lineage. car t cell therapy has become a standard of care treatment for patients with. Abstract. chimeric antigen receptor (car) t cell therapy represents a major advancement in personalized cancer treatment. in this strategy, a patient's own t cells are genetically engineered to express a synthetic receptor that binds a tumor antigen. car t cells are then expanded for clinical use and infused back into the patient's body to.
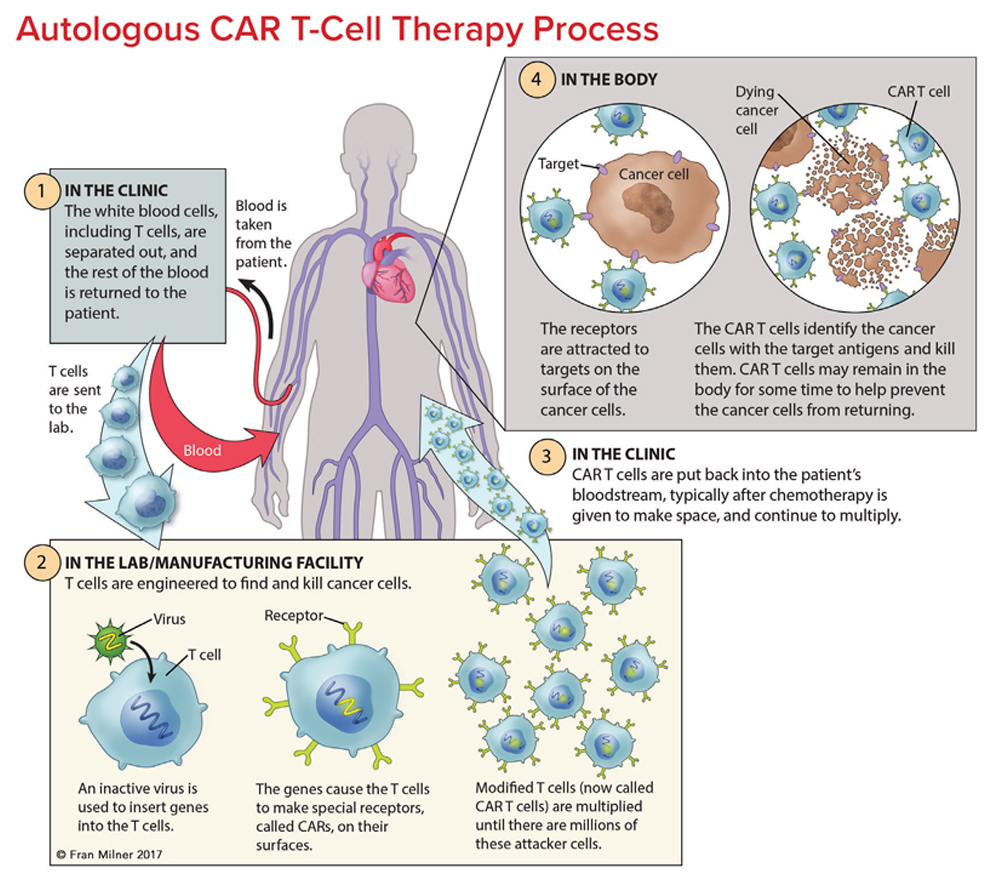
Chimeric Antigen Receptor Car T Cell Therapy 2022 Chimeric antigen receptor (car) t cells are highly effective at targeting and eliminating cells of the b cell lineage. car t cell therapy has become a standard of care treatment for patients with. Abstract. chimeric antigen receptor (car) t cell therapy represents a major advancement in personalized cancer treatment. in this strategy, a patient's own t cells are genetically engineered to express a synthetic receptor that binds a tumor antigen. car t cells are then expanded for clinical use and infused back into the patient's body to. Abstract. chimeric antigen receptor (car) t cells are highly effective at targeting and eliminating cells of the b cell lineage. car t cell therapy has become a standard of care treatment for patients with relapsed or refractory b cell malignancies. in addition, the administration of genetically modified t cells with the capacity to deplete b. Chimeric antigen receptor (car) t cell therapy is a novel and effective immunotherapy strategy for r r hematopoietic malignancies, but relapses can occur due to the loss of car t cells in vivo or.