
Chimeric Antigen Receptor T Cell Therapy For Cancer Clinical Amrolia pj, wynn r, hough r, vora a, bonney d, veys p, et al. simultaneous trgeting of cd19 and cd22: phase i study of auto3, a bicistronic chimeric antigen receptor (car) t cell therapy, in. Cd19 specific chimeric antigen receptor (car) t cells induce high rates of initial response among patients with relapsed b cell acute lymphoblastic leukemia (all) and long term remissions in a subg.

Is Bio Distribution Study Necessary For Car T Therapy вђ Creative Chimeric antigen receptor (car) t cells are engineered fusion proteins that target t cells to a specific antigen present on tumour cells to generate an antitumour immune response 1,2,3.car t cells. Cd19 specific chimeric antigen receptor t cell (cd19 car t) therapy has shown high remission rates in patients with refractory relapsed b cell acute lymphoblastic leukemia (r r b all). however, the long term outcome and the factors that influence the efficacy need further exploration. here we report …. Chimeric antigen receptor (car) t cell therapy is a novel and effective immunotherapy strategy for r r hematopoietic malignancies, but relapses can occur due to the loss of car t cells in vivo or. Autologous chimeric antigen receptor (car) t cell therapy for the treatment of hematologic malignancies has generated intense interest from patients, families and healthcare providers. clinical trials are underway for multiple b cell malignancies, including b cell acute lymphoblastic leukemia (b all), hodgkin lymphoma and non hodgkin lymphoma.
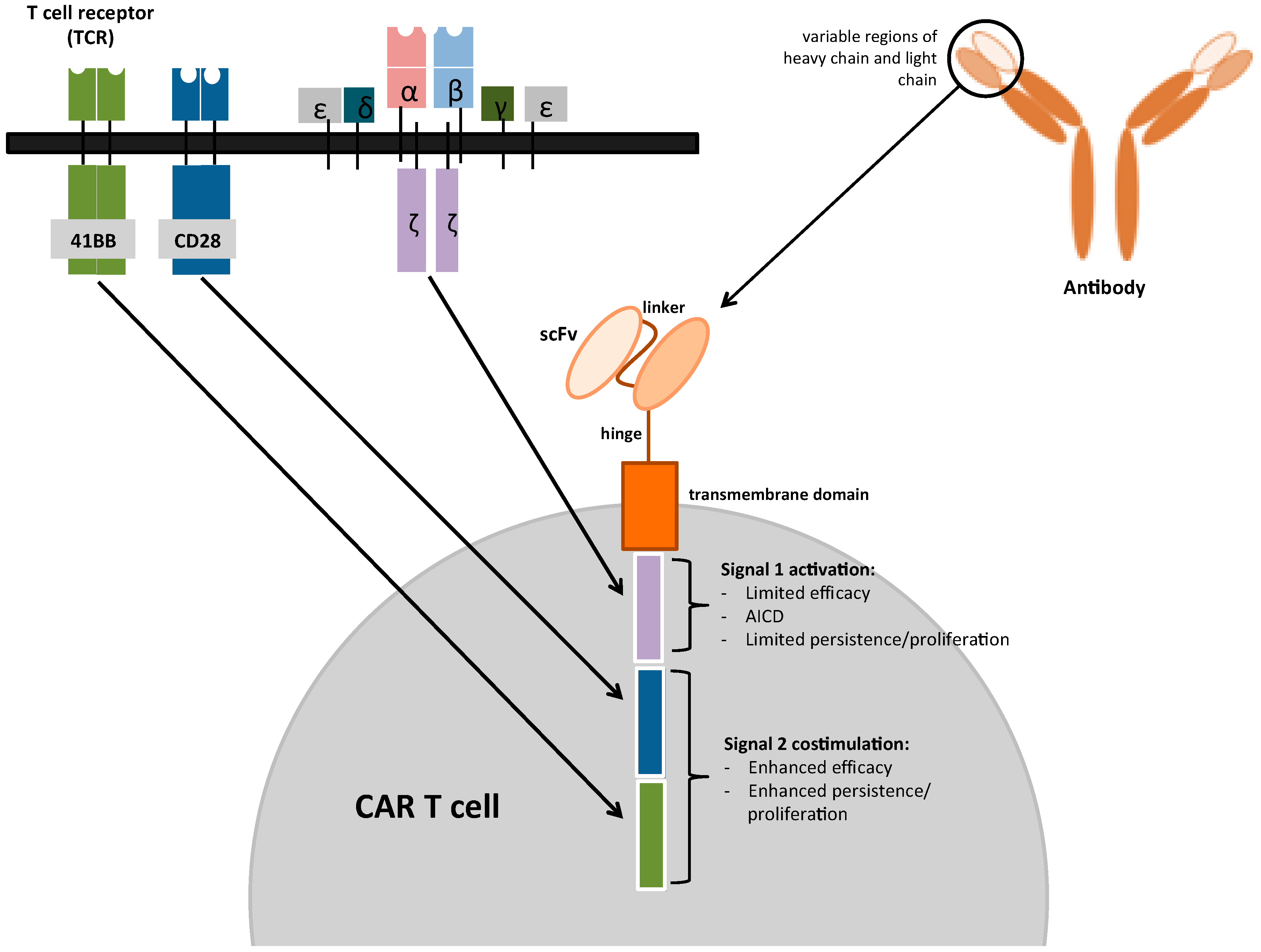
Cancers Free Full Text Chimeric Antigen Receptor Car T Cell Chimeric antigen receptor (car) t cell therapy is a novel and effective immunotherapy strategy for r r hematopoietic malignancies, but relapses can occur due to the loss of car t cells in vivo or. Autologous chimeric antigen receptor (car) t cell therapy for the treatment of hematologic malignancies has generated intense interest from patients, families and healthcare providers. clinical trials are underway for multiple b cell malignancies, including b cell acute lymphoblastic leukemia (b all), hodgkin lymphoma and non hodgkin lymphoma. It is the proof of concept for t cell based immunotherapies in aml based on the graft versus leukemia (gvl) effect, but it also bears the risk of graft versus host disease. cd19 targeting therapies employing chimeric antigen receptor (car) t cells are a breakthrough in cancer therapy. a similar approach for myeloid malignancies is highly desirable. Chimeric antigen receptor t cell (car t) therapy is a form of immunotherapy that has enabled patients with previously incurable hematological cancers to go into remission. currently marketed car t therapies include yescarta, tecartus, kymriah, breyanzi, carvykti, and abecma. indications are acute lymphoblastic leukemia, large b cell lymphoma.
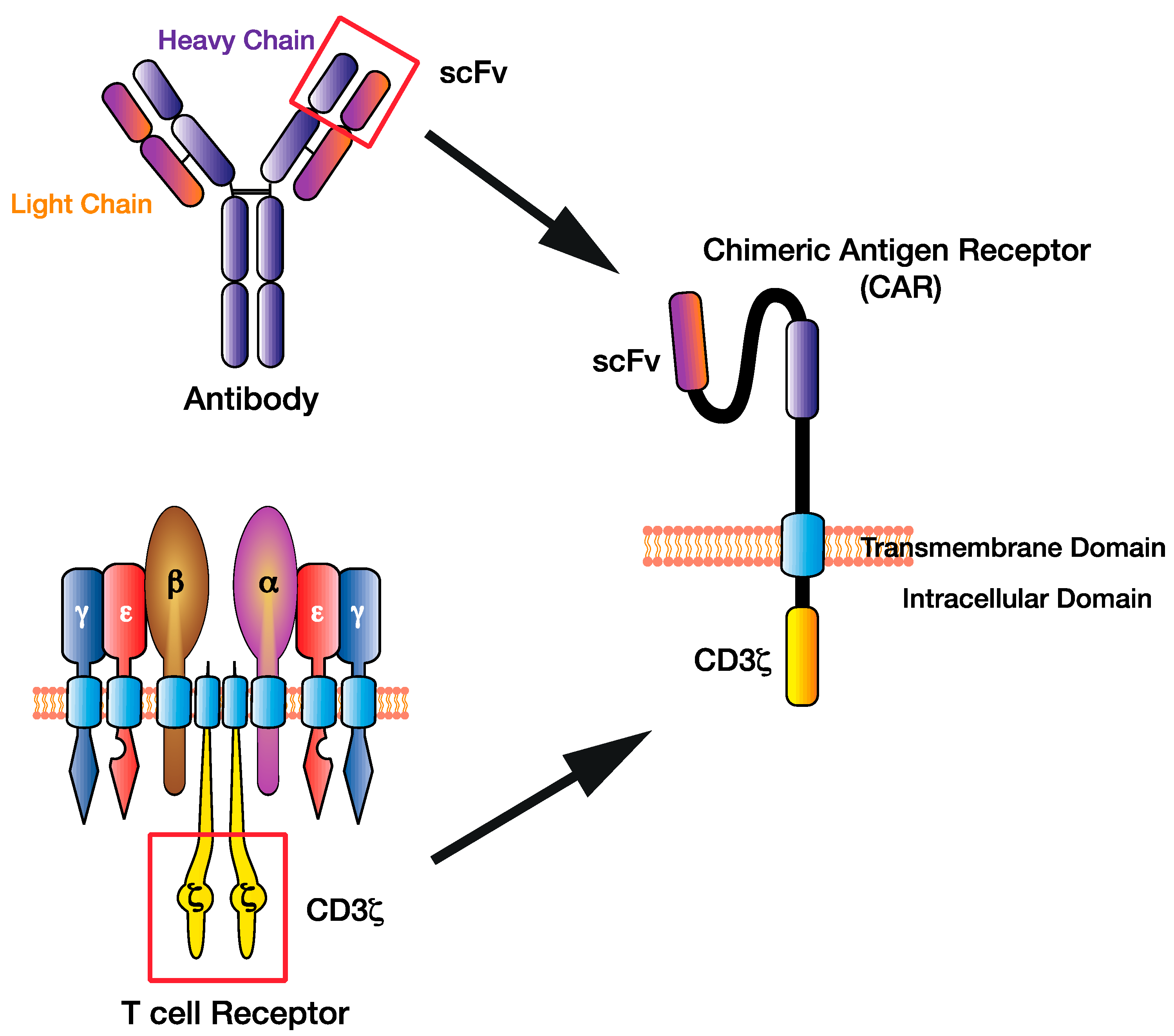
Pdf Chimeric Antigen Receptor Engineered T Cells For Immunotherapy Of It is the proof of concept for t cell based immunotherapies in aml based on the graft versus leukemia (gvl) effect, but it also bears the risk of graft versus host disease. cd19 targeting therapies employing chimeric antigen receptor (car) t cells are a breakthrough in cancer therapy. a similar approach for myeloid malignancies is highly desirable. Chimeric antigen receptor t cell (car t) therapy is a form of immunotherapy that has enabled patients with previously incurable hematological cancers to go into remission. currently marketed car t therapies include yescarta, tecartus, kymriah, breyanzi, carvykti, and abecma. indications are acute lymphoblastic leukemia, large b cell lymphoma.