
Crispr Engineered T Cells In Patients With Refractory Cancer Science The patients were given lymphodepleting chemotherapy with cyclophosphamide and fludarabine on days −5 to −3 (i.e., before administration with crispr cas9–engineered t cells) and a single infusion of 1 × 10 8 manufactured crispr cas9–engineered t cells per kilogram on day 0 of the protocol (fig. s2). no cytokines were administered to. Abstract. crispr cas9 gene editing provides a powerful tool to enhance the natural ability of human t cells to fight cancer. we report a first in human phase 1 clinical trial to test the safety and feasibility of multiplex crispr cas9 editing to engineer t cells in three patients with refractory cancer. two genes encoding the endogenous t cell.

Crispr Engineered T Cells In Patients With Refractory Cancer Science Three patients with advanced, refractory cancer were given infusions of the crispr cas9 engineered t cells. the infusions were well tolerated with no serious adverse events ( table 2 ); importantly there were no cases of cytokine release syndrome, which is a potentially life threatening systemic inflammatory response that has been associated. We report a first in human phase 1 clinical trial to test the safety and feasibility of multiplex. crispr cas9 editing to engineer t cells in three patients with refractory cancer. two genes. Crispr cas9 engineering of t cells in cancer patients. t cells (center) were isolated from the blood of a patient with cancer. crispr cas9 ribonuclear protein complexes loaded with three sgrnas were electroporated into the normal t cells, resulting in gene editing of the trac, trbc1, trbc2, and pdcd1 (encoding pd 1) loci. This is the first u.s. clinical trial to test the gene editing approach in humans, and the publication of this new data today in science follows on the initial report last year that researchers were able to use crispr cas9 technology to successfully edit three cancer patients’ immune cells. read the department of communications news release.
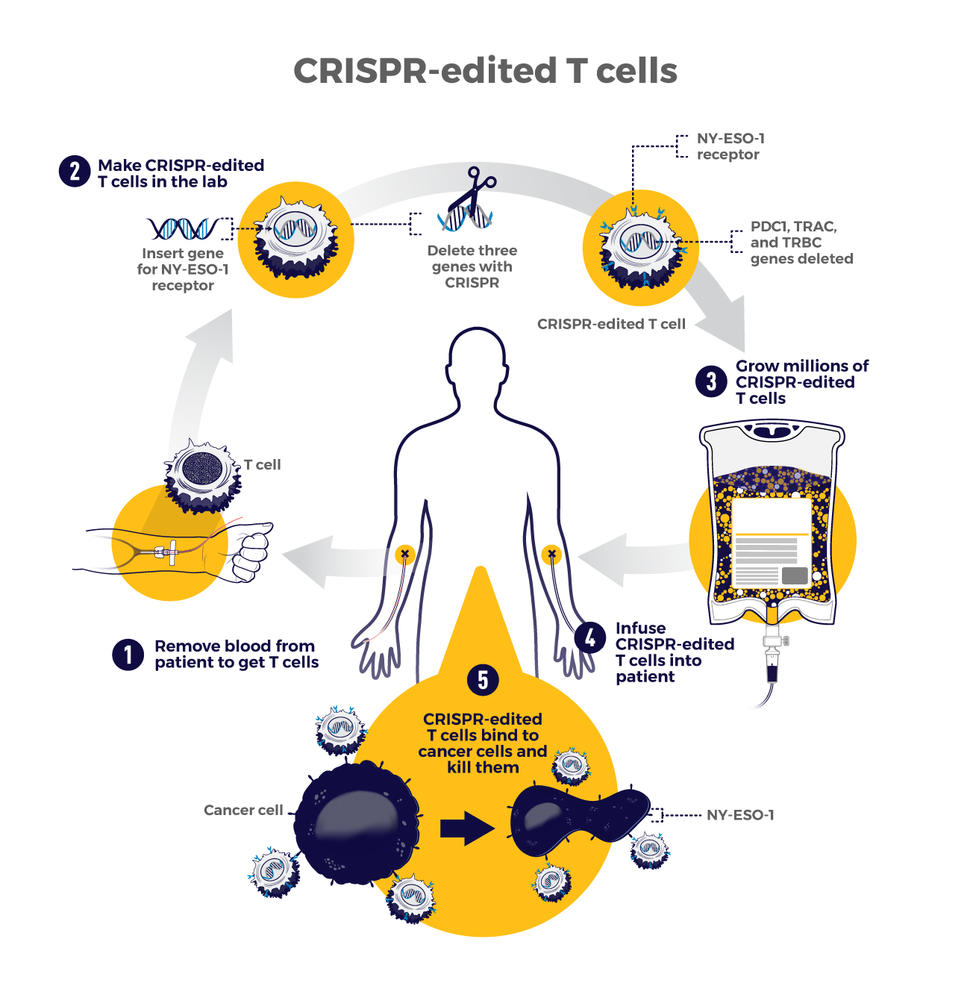
How Is Crispr Used In Cancer Research Molecularcloud Crispr cas9 engineering of t cells in cancer patients. t cells (center) were isolated from the blood of a patient with cancer. crispr cas9 ribonuclear protein complexes loaded with three sgrnas were electroporated into the normal t cells, resulting in gene editing of the trac, trbc1, trbc2, and pdcd1 (encoding pd 1) loci. This is the first u.s. clinical trial to test the gene editing approach in humans, and the publication of this new data today in science follows on the initial report last year that researchers were able to use crispr cas9 technology to successfully edit three cancer patients’ immune cells. read the department of communications news release. Chimeric antigen receptor (car) t cells represent a breakthrough in personalized cancer therapy. in this strategy, synthetic receptors comprised of antigen recognition, signaling, and costimulatory domains are used to reprogram t cells to target tumor cells for destruction. despite the success of this approach in refractory b cell malignancies, optimal potency of car t cell therapy for many. T cell receptors (tcrs) enable t cells to specifically recognize mutations in cancer cells1–3. here we developed a clinical grade approach based on crispr–cas9 non viral precision genome.

Crispr Engineered T Cells In Patients With Refractory Cancerо Chimeric antigen receptor (car) t cells represent a breakthrough in personalized cancer therapy. in this strategy, synthetic receptors comprised of antigen recognition, signaling, and costimulatory domains are used to reprogram t cells to target tumor cells for destruction. despite the success of this approach in refractory b cell malignancies, optimal potency of car t cell therapy for many. T cell receptors (tcrs) enable t cells to specifically recognize mutations in cancer cells1–3. here we developed a clinical grade approach based on crispr–cas9 non viral precision genome.