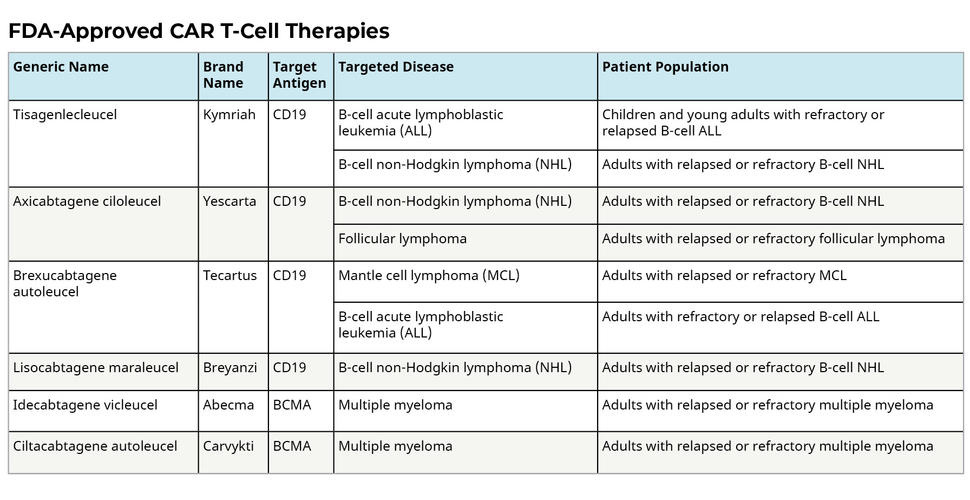
Car T Cells Engineering Immune Cells To Treat Cancer 2022 The field of car t cell therapy is then continually developing, with an explosion of over a thousand clinical trials on car t therapies globally to cure advanced cancers that are resistant or refractory to the conventional therapies. 174 while some of these clinical trials have shown promising outcomes in cancer patients, many other car t cells. Chimeric antigen receptor (car) t cell therapy is a novel, customized immunotherapy that is considered a 'living' and self replicating drug to treat cancer, sometimes resulting in a complete cure. car t cells are manufactured through genetic engineering of t cells by equipping them with cars to detect and target antigen expressing cancer cells.
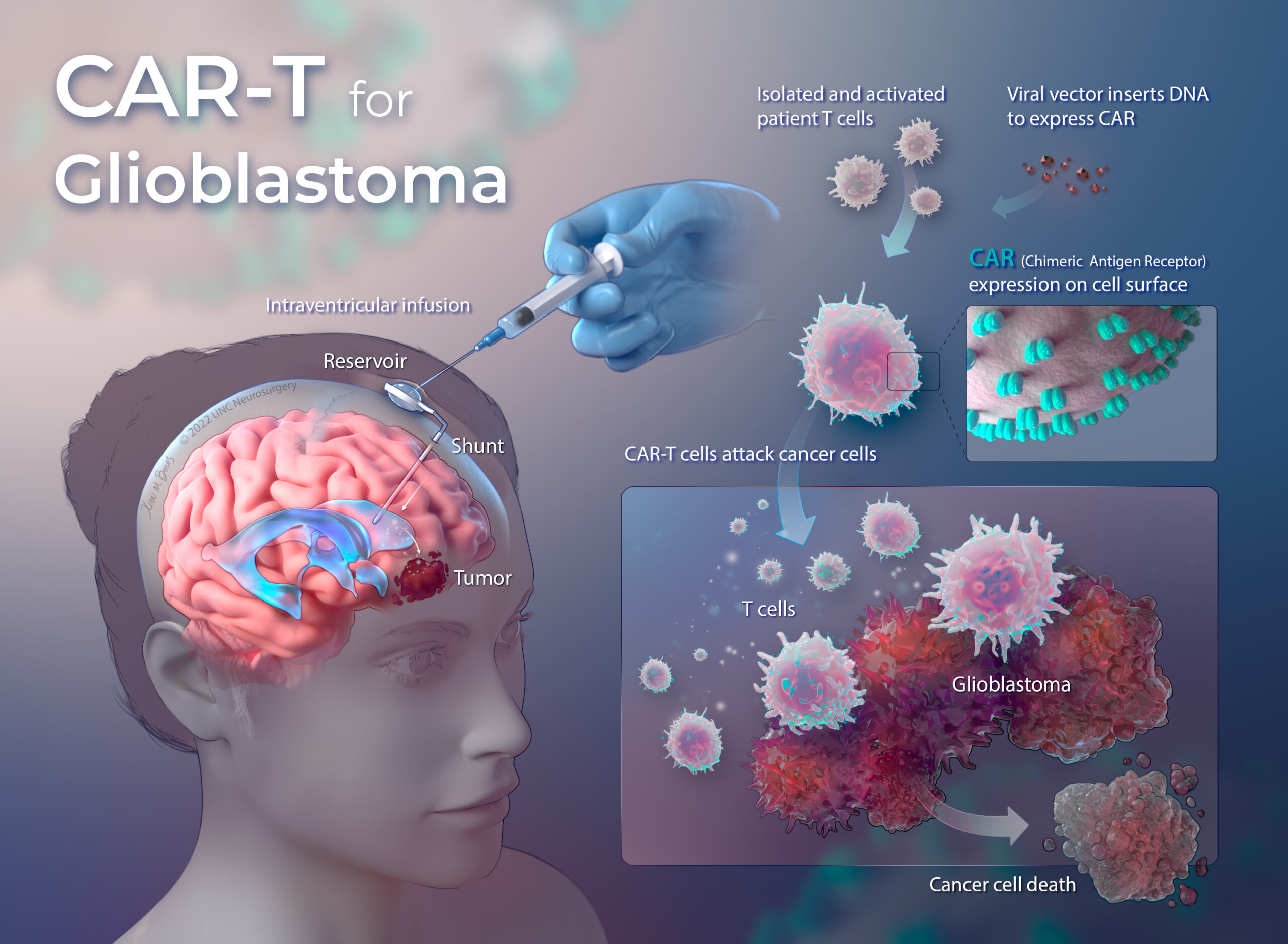
Phase I Clinical Trial Using Car T For Glioblastoma To Begin At Unc In total, over 500 clinical trials analyzing car t cells for the treatment of cancer are currently being conducted around the world. thereof, the majority are being performed in east asia (269. The development of car t cell clinical trials has accelerated over the last two decades. these trials, collected on clinicaltrial.gov until 2022, come mainly from the united states ( n = 377) and china ( n = 636), while europe ( n = 58) remains far behind these two leading countries. the aim of our analysis of clinical trials was to provide an. Next generation car t therapies. the innovation landscape for car t therapies can be classified across two axes: cell sourcing and engineering approaches (fig. 1). with the progression from. This means that eradicating bcma expressing cells using car t tcr t approach is a sound therapeutic option. we have identified 85 clinical trials that target bcma, and car t clinical trials targeting this antigen are the fastest growing after cd19. current clinical trial pipelines consist of over 40 different bcma car t programs.
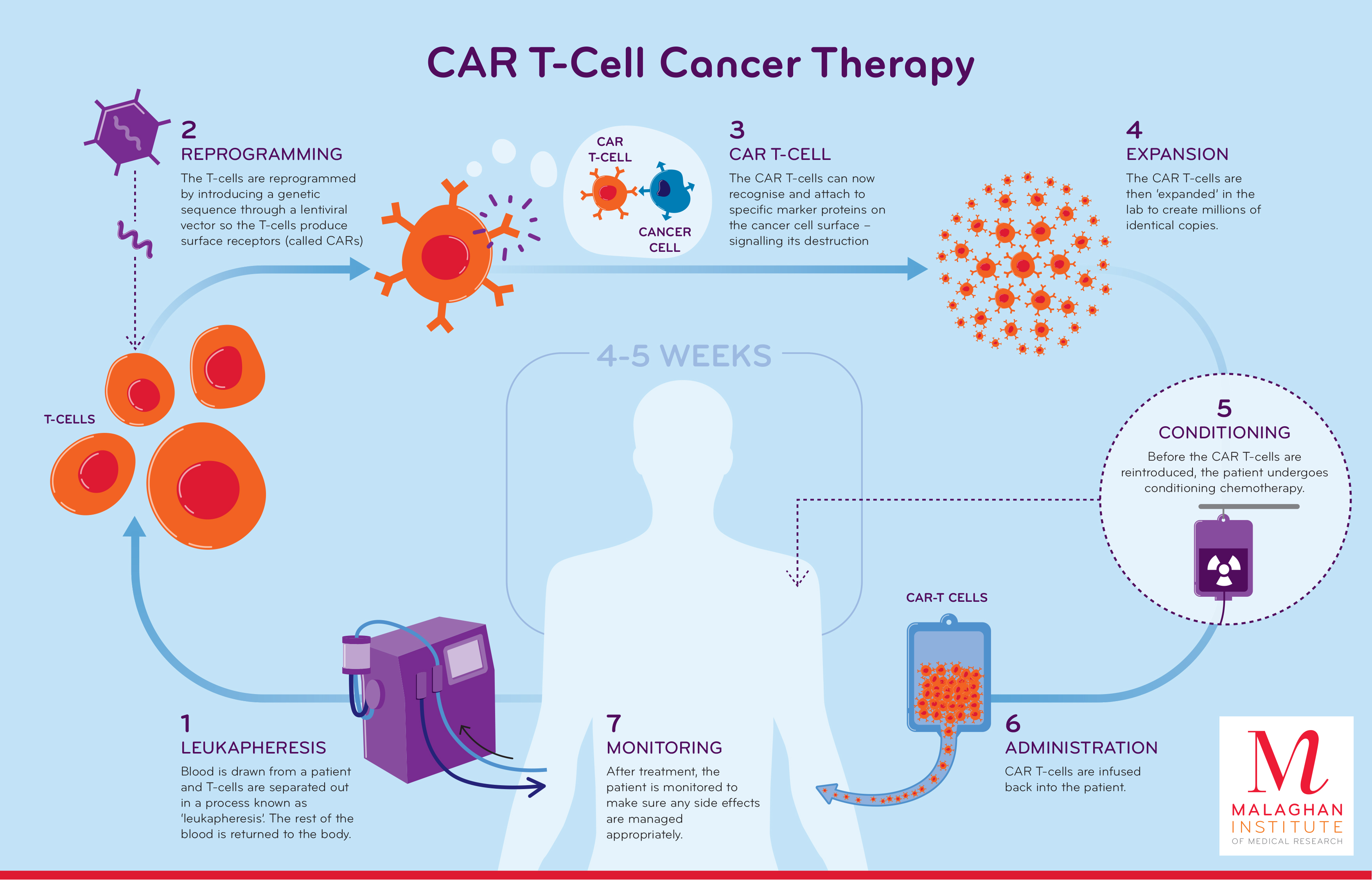
Car T Cell Therapy Next generation car t therapies. the innovation landscape for car t therapies can be classified across two axes: cell sourcing and engineering approaches (fig. 1). with the progression from. This means that eradicating bcma expressing cells using car t tcr t approach is a sound therapeutic option. we have identified 85 clinical trials that target bcma, and car t clinical trials targeting this antigen are the fastest growing after cd19. current clinical trial pipelines consist of over 40 different bcma car t programs. Cd19 directed car t cell therapy for the treatment of relapsed refractory b cell malignancies. rochester, minn. the purpose of this study is to find out more about the side effects of the car t therapy called ic19 1563 and what dose of ic19 1563 is safe for patients. the therapy, ic19 1563, uses some of the patients own immune cells, called t. Ongoing clinical trials on car t cell therapy. many years ago, the first car t cell clinical trial began by targeting the folate receptor to treat patients with advanced epithelial ovarian cancer. citation 143 the breakthrough, however, came in the subsequent years with cd19 targeted car t cells used to treat b cell malignancies. the field of.
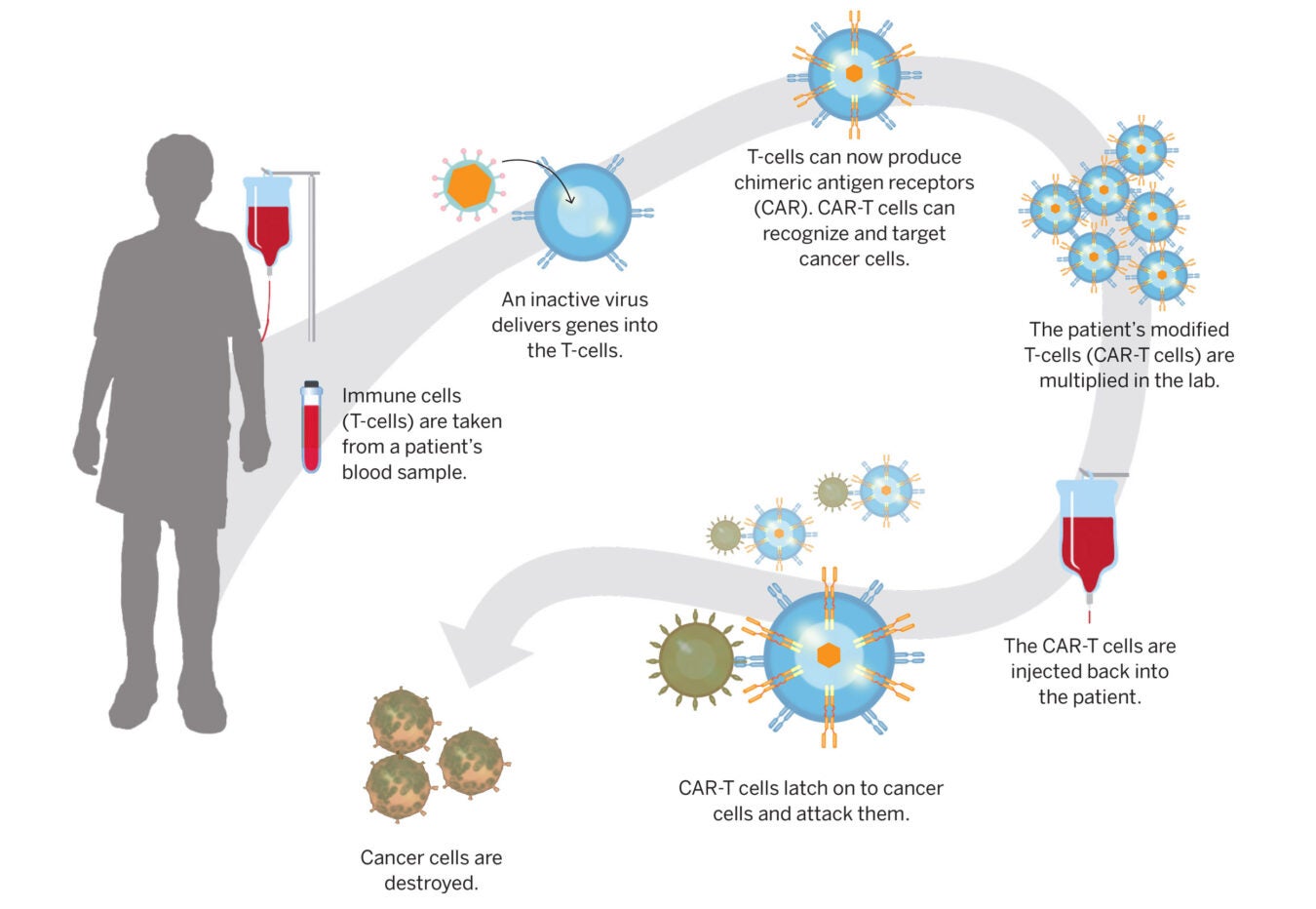
Partnership Aims To Accelerate Cell And Gene Therapy вђ Harvard Gazette Cd19 directed car t cell therapy for the treatment of relapsed refractory b cell malignancies. rochester, minn. the purpose of this study is to find out more about the side effects of the car t therapy called ic19 1563 and what dose of ic19 1563 is safe for patients. the therapy, ic19 1563, uses some of the patients own immune cells, called t. Ongoing clinical trials on car t cell therapy. many years ago, the first car t cell clinical trial began by targeting the folate receptor to treat patients with advanced epithelial ovarian cancer. citation 143 the breakthrough, however, came in the subsequent years with cd19 targeted car t cells used to treat b cell malignancies. the field of.

Personalized Cell Therapy Market Will Continue To Boom In The World Of