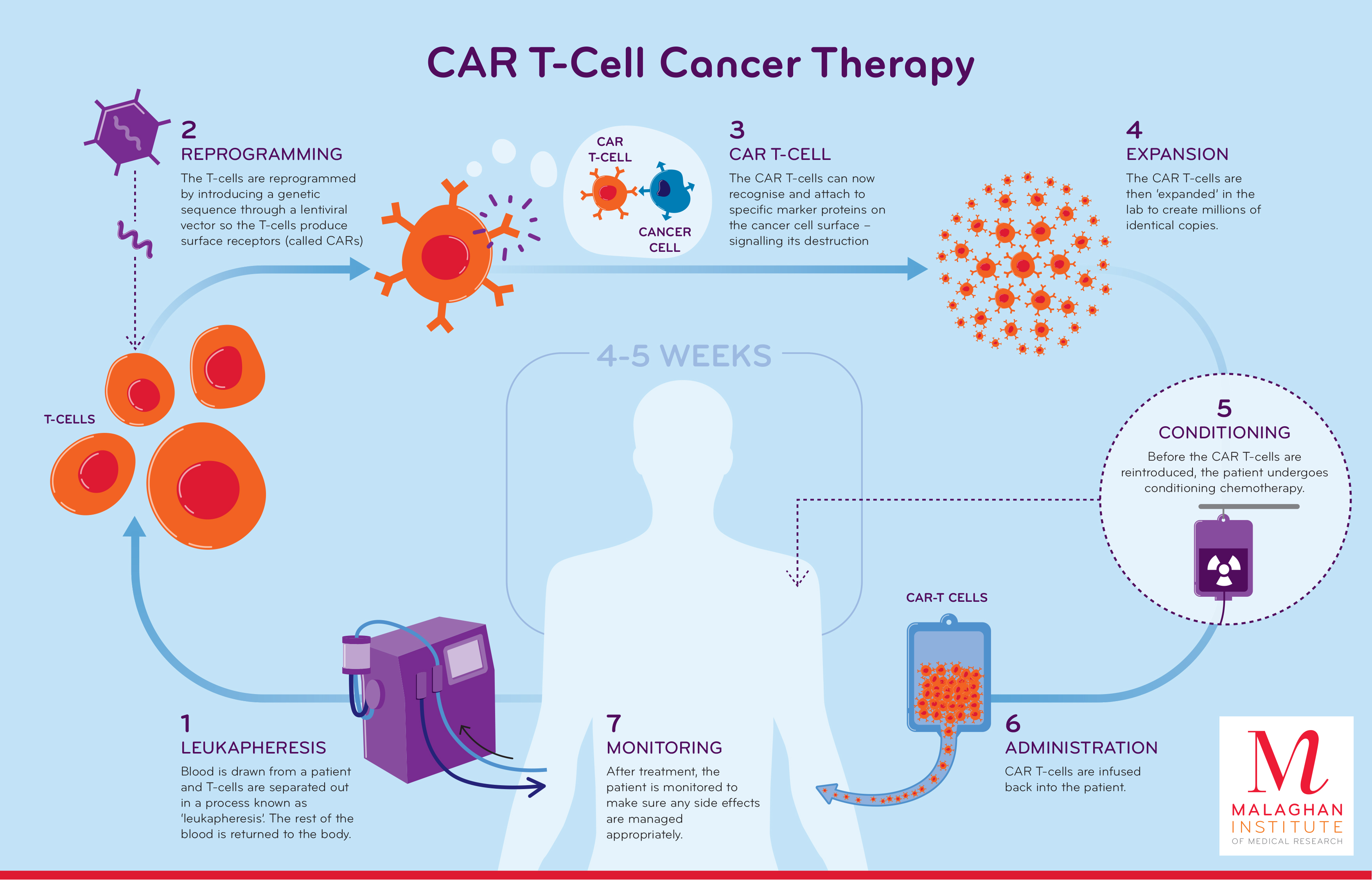
Car T Cell Therapy Process My Xxx Hot Girl The application of car t in disease treatment. november 22, 2017 chimeric antigen receptor research, tcr research car t, tcr. chimeric antigen receptor (car) modified t cells (car t) as a new field of adoptive immune cell therapy has become a hot spot in recent research. in 1989, israeli scholar gross et al. first proposed the concept of car t. As a precision targeted therapy for tumor treatment, car t (chimeric antigen receptor t cell immunotherapy) has achieved promising results in clinical tumor treatment with optimization and improvement in recent years. it is cutting edge tumor immunotherapy that is accurate, fast, efficient, and has the potential to cure cancer.

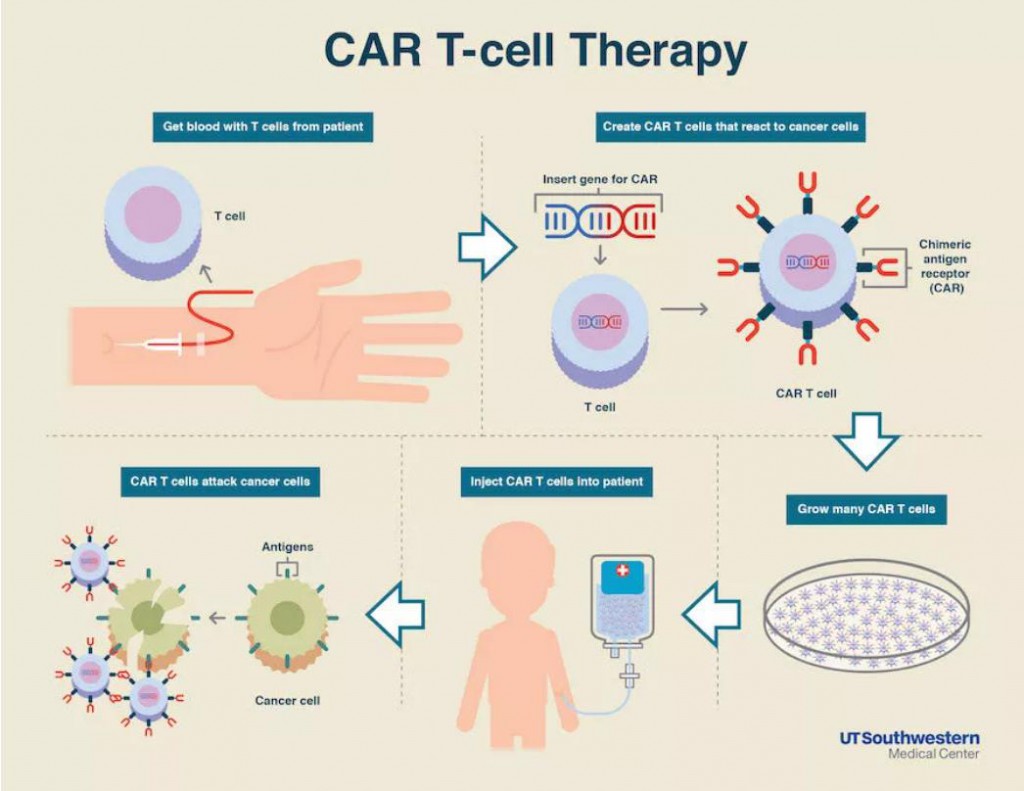
Antibody Based Therapy вђ Creative Biolabs Blog Car t for blastoma therapy. a blastoma refers to the tumor that arises from embryonic tissue, typical use childhood cancer. the term blastoma is generally included as part of the full medical name of a specific tumor, as in medulloblastoma, neuroblastoma, or pleuropul blast blastoma. immunotherapy of adoptive t cells provides a very promising. Chimeric antigen receptor (car) t cell therapy stands out from traditional cancer treatments, such as nonspecific drugs and monoclonal antibodies, by employing the immune system's t cells to directly recognize and eliminate tumor cells. car t cell therapy involves isolating t cells from a patient, genetically modifying them to express cars that. This infographic presents clinical applications and challenges in car t cell therapies, including approved car t therapies, representative car t therapies in development, and challenges and countermeasures. On july 9th, fate therapeutics announced that u.s. food and drug administration has approved the first ind application for ipsc derived allogeneic car t cell therapy ft819 for the treatment of recurrent refractory b cell malignant tumors, including chronic lymphoblastic leukemia (cll), acute lymphoblastic leukemia (all) and non hodgkin’s lymphoma (nhl).
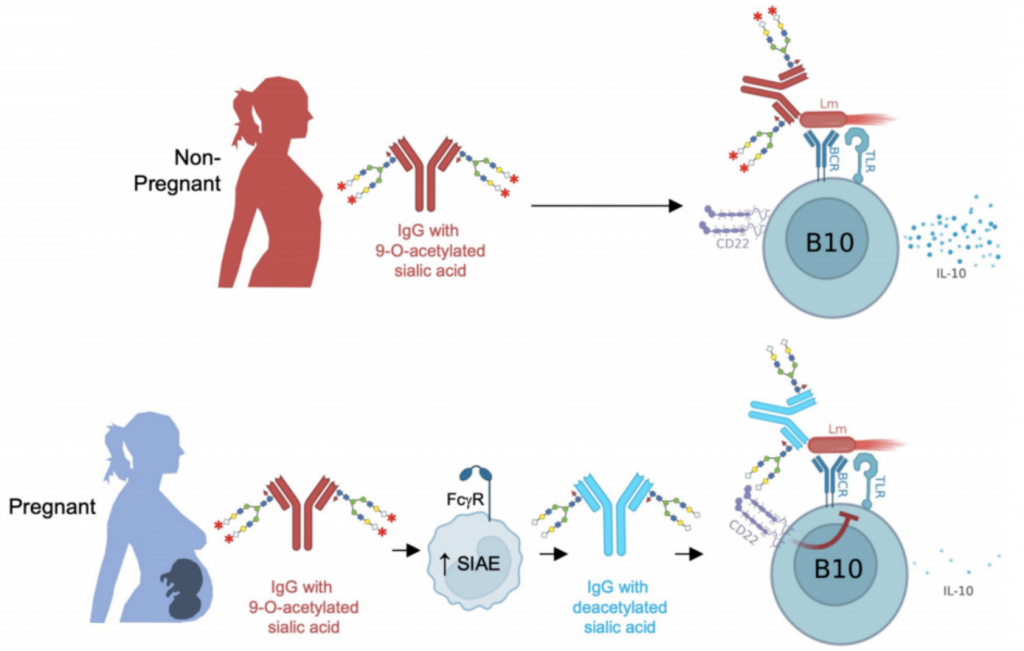
Pregnant Women Produce Super Antibodies To Protect Babies Against This infographic presents clinical applications and challenges in car t cell therapies, including approved car t therapies, representative car t therapies in development, and challenges and countermeasures. On july 9th, fate therapeutics announced that u.s. food and drug administration has approved the first ind application for ipsc derived allogeneic car t cell therapy ft819 for the treatment of recurrent refractory b cell malignant tumors, including chronic lymphoblastic leukemia (cll), acute lymphoblastic leukemia (all) and non hodgkin’s lymphoma (nhl). Engineered γδ t cells, for example, combined with car technology, should be considered as a new platform for adoptive t cell cancer therapy. creative biolabs offers our perspectives and services to help make γδ t cells promising for use in immunotherapy against several types of solid tumors, such as melanoma and gastrointestinal cancers. With extensive industrial experience, creative biolabs provides high quality car engineered γδ t cell functional test services to global customers. fig.1 functional advantages of γδ t cells for car t cell cancer therapy. (mirzaei, 2016) services at creative biolabs the functional test of γδ t cells is a systematical process.
Carl June The Worldтащs First юааcarюаб юааtюаб Therapy Released The Latest Data Engineered γδ t cells, for example, combined with car technology, should be considered as a new platform for adoptive t cell cancer therapy. creative biolabs offers our perspectives and services to help make γδ t cells promising for use in immunotherapy against several types of solid tumors, such as melanoma and gastrointestinal cancers. With extensive industrial experience, creative biolabs provides high quality car engineered γδ t cell functional test services to global customers. fig.1 functional advantages of γδ t cells for car t cell cancer therapy. (mirzaei, 2016) services at creative biolabs the functional test of γδ t cells is a systematical process.